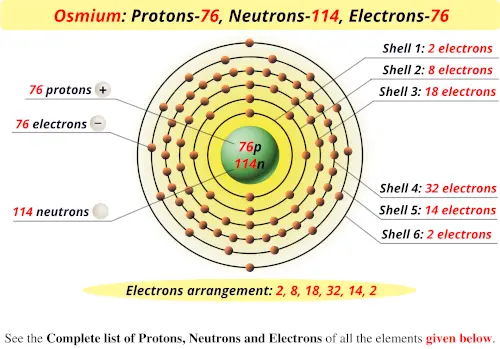
Graphical depiction of the electron cloud for the helium (He) atom. 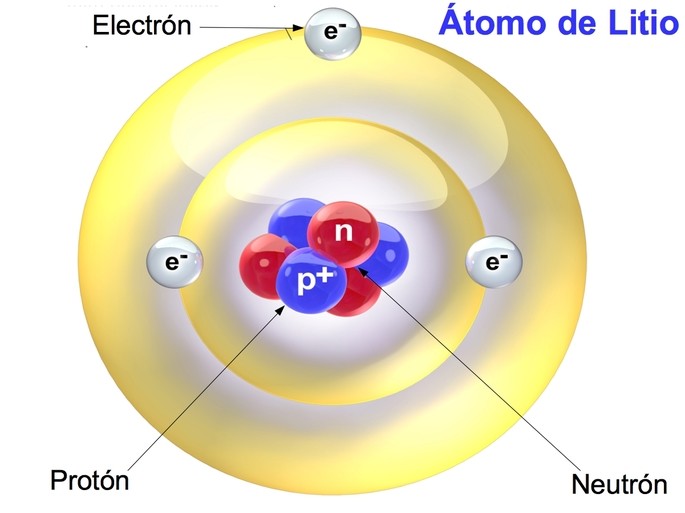
More details about the energies and distributions of electrons in the atom will be discussed in a later module on light, matter, and atomic structure. We use the term “electron cloud” because the exact positions of the electrons cannot be known, we simply know that they are distributed about the volume of the atom ( Figure 2). 
Most of the volume of an atom is occupied by the “cloud” of electrons. (credit middle: modification of work by “babyknight”/Wikimedia Commons credit right: modification of work by Paxson Woelber) Their diameters differ by about 20,000-60,000 times. If an atom could be expanded to the size of a football stadium, the nucleus would be the size of a single blueberry. A third subatomic component that bears no electrical charge-the neutrons (n 0)-have masses similar to protons and co-locate with protons in the nucleus.įor a perspective about their relative sizes, consider this: if the nucleus were the size of a blueberry, the atom would be about the size of a football stadium ( Figure 1). The surprising results of Rutherford’s experiments, which we will explore in more detail in the next section, indicated that the positively charged subatomic particles-the protons (p +)-are much more massive than electrons (by a factor of about 2,000) and are highly concentrated in the atom’s nucl e us, which occupies just a tiny portion of the atom’s entire volume. We designate electrons with the symbol “e –“, the superscript negative sign emphasizes their negative charge. The work of Thomson, Millikan, and others revealed the charge and mass of the negative subatomic particles-the electrons. Much of our current understanding of atoms and atomic structure were developed in the early part of the 20 th century. | Key Concepts and Summary | Glossary | End of Section Exercises | The Structure of the Atom Know the names and symbols of certain elements.Recognize the atom is comprised of protons, neutrons, and electrons distinguish among these building blocks.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
